Call us Today! 877-722-8910
Meet the CLC Family of Clinical Chemistry Analyzers
Instruments and Reagents for Any Size Chemistry Lab
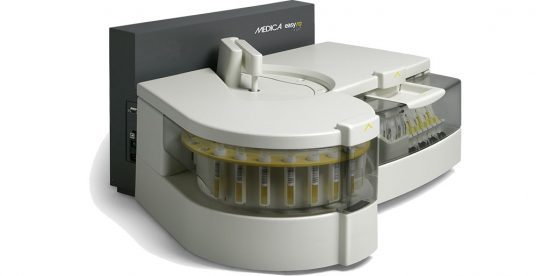
Fully Automated, Open System Benchtop Clinical Chemistry Analyzer
Throughput Up to 270 tests/hr
Designed for Large Hospitals and Reference Labs
Throughput
Up to 1600 tests/hr per module
Up to 960 tests/hr on ISE module
Moderately Complex General Chemistries and Urine Drug Screening on One System
Throughput
Up to 300 tests/hr, 8 min STAT
40 General Chemistries
27 Urine Drug Screens
15 Immunochemistries
24 Special Chemistries, including:
- Lp-PLA2 (PLAC®)
- Vitamin D (2-part)
- Procalcitonin (PCT)
- Human Κ/λ Free Light Chain
Waived (FDA EUA Granted)
Waived, Point-of-Care (POC)
(FDA EUA Granted)
COVID-19 Testing Supplies
Chemistry analyzers and reagents for clinical laboratories of all sizes
Carolina Liquid Chemistries offers industry-leading clinical chemistry analyzers, testing reagents and support/service for all sizes of clinical laboratories: physician, hospital and reference.

Chemistry Analyzers
Our leading-edge lines of clinical chemistry analyzers for all sizes of laboratories

Reagents & Service
A complete line of general chemistry and toxicology (urine drug screening) reagents, support and service

Refurbished Products
Affordable, refurbished Olympus AU and BioLIS 24i/CLC480 clinical chemistry analyzers for laboratories